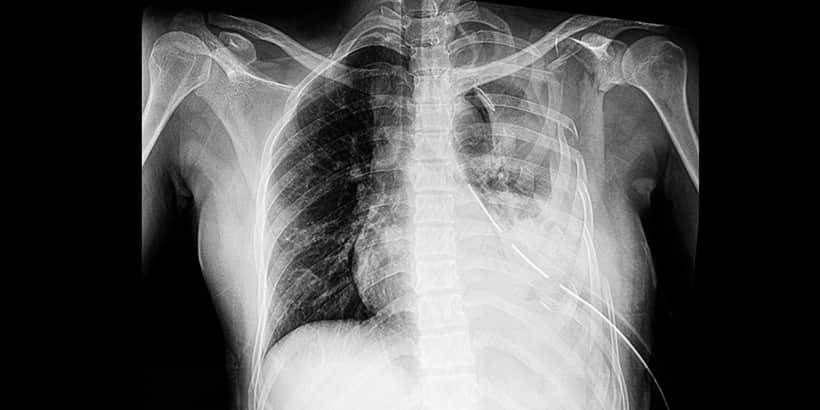
Pneumothorax
 The pleural layer in the chest is a serous membrane that lines the thoracic wall and lungs and can be divided into two types. The parietal layer is attached to the chest wall, diaphragm, and mediastinum. The visceral layer is an extension of the parietal layer that has been reflected back to cover the lungs. These two layers are very close together with a small amount of parietal fluid in the pleural space that separates them. If communication is created across the pleura, accumulation of air in the pleural space is referred to as a pneumothorax.12
The pleural layer in the chest is a serous membrane that lines the thoracic wall and lungs and can be divided into two types. The parietal layer is attached to the chest wall, diaphragm, and mediastinum. The visceral layer is an extension of the parietal layer that has been reflected back to cover the lungs. These two layers are very close together with a small amount of parietal fluid in the pleural space that separates them. If communication is created across the pleura, accumulation of air in the pleural space is referred to as a pneumothorax.12
Three subdivisions exist when categorizing a pneumothorax, depending on whether air has direct access to the pleural cavity.
Simple Pneumothorax
A simple pneumothorax has no communication between the atmosphere and the pleural space.11 The severity of the pneumothorax is graded on the basis of degree of collapse: collapse of 15% or less is small; collapse of 15% to 60% is moderate; and collapse of greater than 60% is large.12
Communicating Pneumothorax
With a communicating pneumothorax, air in the pleural cavity exchanges with atmospheric air through a defect in the chest wall.11 A communicating pneumothorax represents a severe respiratory disturbance because the affected lung collapses on inspiration and expands slightly on expiration.12
Tension Pneumothorax
A tension pneumothorax develops when air progressively accumulates under pressure within the pleural cavity.11 If the pressure becomes too great, the mediastinum shifts to the opposite hemithorax, and this causes compression of the contralateral lung and great vessels.12
Primary versus Secondary Pneumothorax
Primary spontaneous pneumothorax (PSP) conventionally refers to patients with no underlying lung disease, while those with established lung pathology are classified as secondary spontaneous pneumothorax (SSP).6 Spontaneous pneumothorax is a relatively common finding in emergency departments, occurring primarily in adolescents and young adult males.10 Secondary spontaneous pneumothorax more commonly affects the elderly and is most frequently observed in patients with chronic obstructive pulmonary disease (COPD), but also with interstitial lung disease, pneumonia, malignancies, and other disorders.7
Treatment
Treatment for a simple pneumothorax depends on the size and the resulting effect it is having on the patient’s respiratory/cardiovascular status. Close observation is warranted to determine if needle aspiration or tube thoracostomy is indicated. According to Sahota et al, the reasons to not place any kind of drain are:
- Small to moderate-sized pneumothorax.
- Some types of pneumothorax can resolve spontaneously; patients are very unlikely to not deteriorate clinically if monitored when undergoing conservative management.
A communicating pneumothorax needs rapid intervention to prevent worsening complications. The area of injury should be covered with an occlusive dressing to prevent the influx of air, but allow the egress of air from inside the chest cavity.12 By allowing the air to escape, the pneumothorax does not get any larger and the pressure can’t build and transform the injury into a tension pneumothorax.
Tension pneumothorax is a life-threating process that needs emergent treatment. The recommended first-line treatment of suspected tension pneumothorax according to the Advanced Trauma Life Support (ATLS) and Pre-Hospital Trauma Life Support (PHTLS) guidelines is needle thoracentesis (NT) in the second intercostal space (2ICS)1. Although widely recommended, the insertion site in 2ICS is not based on solid evidence (Fitzgerald). Unsuccessful needle decompression may be attributable to insufficient needle length, inaccurate needle positioning, catheter displacement, plugging of the catheter lumen, and kinking due to the small diameter of the catheter.5
As an alternative to the 2ICS, it has been demonstrated that the chest wall is approximately 1.3 cm thinner at the fifth ICS in the anterior axillary line (5ICS).8 Decompression in the 5ICS is performed in the mid-anterior axillary line, posterior to the major pectoral muscle. Incision should be no lower than the level of the nipple in males and the inframammary crease in females.9 A rush of air is heard with successful placement of the needle thoracentesis and when the angiocatheter used for NT converts the tension pneumothorax into a simple pneumothorax.
Inaccurate needle insertion increases the risk of complications, especially intravascular insertion.4 Due to the risk of failed decompression using NT, several authors recommend lateral thoracostomy in the hemodynamic unstable trauma patient.5 The procedure is performed using a sharp incision through the skin in the anterior axillary line of 3rd–5th ICS.2 This is followed by blunt dissection to the parietal pleura with a finger or a blunt dissecting instrument (e.g., Kelly clamp).2 The pleura should be penetrated by a finger and the incision left open until a definite drain can be placed through the same incision.2
Sahota et al. listed the following reasons to place a chest tube:
- If the pneumothorax is initially >1.5 cm in size, the patient needs a chest drain insertion.
- If the patient is hemodynamically unstable or has a multitude of other injuries, the patient needs a chest drain.
- If the patient requires IPPV (intermittent positive pressure ventilation), a chest drain is always required, due to increased risk of a tension pneumothorax developing.
- If the patient is symptomatic from the traumatic pneumothorax, especially if they are older.
- Only 10% of patients initially managed conservatively then go on to have a drain inserted.
The primary concern is the immediate decompression; however, in the spontaneous breathing patient, a chest tube should be placed immediately after decompression to allow lung expansion hemodynamically.2 Insertion of a chest tube is the preferred treatment of the pneumothorax, and both surgical and Seldinger techniques are both acceptable methods of chest drain insertion.3 The lateral thoracostomy using blunt dissection is a safe procedure with a high success rate. It should be considered as the first-line treatment of tension pneumothorax in hospital and pre-hospital, particularly in the unstable patient.2
References
- American College of Surgeons. Committee on Trauma. Pre-hospital trauma life support. 9th ed. American College of Surgeons. Chicago, IL. 2014.
- Bach PT, Sølling C. Failed needle decompression of bilateral spontaneous tension pneumothorax. Acta Anaesthesiologica Scandinavica. 2015.
- Doelken P. Placement and management of thoracostomy tubes. Parsons PE ed. https://www.uptodate.com/contents/placement-and-management-of-thoracostomy-tubes-and-catheters-in-adults-and-children. Accessed October 2, 2014.
- Ferrie EP, Collum N, McGovern S. The right place in the right space? Awareness of site for needle thoracocentesis. Emergency Medicine Journal. 2005;22:788-9.
- Fitzgerald M, Mackenzie CF, Marasco S, Hoyle R, Kossmann T. Pleural decompression and drainage during trauma reception and resuscitation. Injury. 2008;39:9-20.
- Hallifax RJ, Rahman NM. Epidemiology of pneumothorax—finally something solid out of thin air. Thorax. 2015;(70)10:921-922.
- Ichinose J, Nagayama K, Hino H, et al. Results of surgical treatment for secondary spontaneous pneumothorax according to underlying diseases. European Journal of Cardiothoracic Surgery. 2016;49:1132-6.
- Inaba K, Ives C, McClure K, Branco BC, Eckstein M, Shatz D, Martin MJ, Reddy S, Demetriades D. Radiologic evaluation of alternative sites for needle decompression of tension pneumothorax. The Archives of Surgery. 2012;147:813-8.
- Kwiatt M, Tarbox A, Seamon MJ, Swaroop M, Cipolla J, Allen C, Hallenbeck S, Davido HT, Lindsey DE, Doraiswamy VA, Galwankar S, Tulman D, Latchana N, Papadimos TJ, Cook CH, Stawicki SP. Thoracostomy tubes: a comprehensive review of complications and related topics. International Journal of Critical Illness and Injury Science. 2014;4:143-55.
- Lee SC, Cheng YL, Huang CW, Tzao C, Hsu HH, Chang H. Simultaneous bilateral primary spontaneous pneumothorax. Respirology. 2008;13:145-8.
- Marx JA, et al. Rosen’s Emergency Medicine: Concepts and Clinical Practice. 7th ed. Philadephia, PA. 2014:437.
- Nagelhout J J, Elisha S, Rieker M. Nurse Anesthesia. St. Louis, MO. 2019.