Therapeutic Hypothermia
 Cardiac arrest (CA) is associated with very high mortality and causes neurological dysfunction in the survivors.4 Out-of-hospital cardiac arrest (OHCA) affects approximately 300,000 Americans every year.18 CA is a devastating condition and associated with a mortality of more than 90%.15 Although the in-hospital rate of survival for individuals hospitalized following OHCA improved by nearly 12% from 2001 to 2009, the in-hospital mortality rate remains close to 60%.21 Even after the successful return of spontaneous circulation (ROSC) following an OHCA, approximately 80% of patients remain comatose5 and the majority of deaths following the return of spontaneous circulation (ROSC) are attributable to neurological injury.14 To suffer a CA can be a lifechanging experience with a significant impact on one’s day-to-day life.13 Many of the individuals who do survive have a wide range of subsequent neurologic impairments.20 It has been shown that even a short CA of only a few minutes in duration can change the affected individual permanently, including diminished cognitive function regarding emotions and memories.23
Cardiac arrest (CA) is associated with very high mortality and causes neurological dysfunction in the survivors.4 Out-of-hospital cardiac arrest (OHCA) affects approximately 300,000 Americans every year.18 CA is a devastating condition and associated with a mortality of more than 90%.15 Although the in-hospital rate of survival for individuals hospitalized following OHCA improved by nearly 12% from 2001 to 2009, the in-hospital mortality rate remains close to 60%.21 Even after the successful return of spontaneous circulation (ROSC) following an OHCA, approximately 80% of patients remain comatose5 and the majority of deaths following the return of spontaneous circulation (ROSC) are attributable to neurological injury.14 To suffer a CA can be a lifechanging experience with a significant impact on one’s day-to-day life.13 Many of the individuals who do survive have a wide range of subsequent neurologic impairments.20 It has been shown that even a short CA of only a few minutes in duration can change the affected individual permanently, including diminished cognitive function regarding emotions and memories.23
Therapeutic hypothermia (TH) is recommended in post-CA patients who remain comatose after successful resuscitation. TH for patients who remain comatose following resuscitation from a cardiac arrest improves both survival and neurologic outcomes.21 A decrease in metabolism in the brain from TH may be one of the most important mechanisms of neuroprotection.4 Since 2003, International Liaison Committee on Resuscitation (ILCOR) recommends TH treatment for everyone with CA who regains spontaneous circulation.22 TH is now a component of post-cardiac arrest care as per current advanced cardiac life support protocol by American Heart Association/American College of Cardiology.19 A Cochrane review released in 2012 also reported the benefits of TH.2
TH has been defined as a body temperature of below 34°C.4 Differences of opinion exist as to what the goal temperature should be, when should TH be initiated, which CA victims should have the treatment, and so on. Because the pathophysiology for neurologic injury following CA is similar regardless of rhythm or location of arrest, the decision to initiate TH should be made on an individual patient basis considering the etiology of the arrest, time before initiation of CPR, duration of CPR prior to ROSC, and overall prognosis based on underlying comorbidities.21
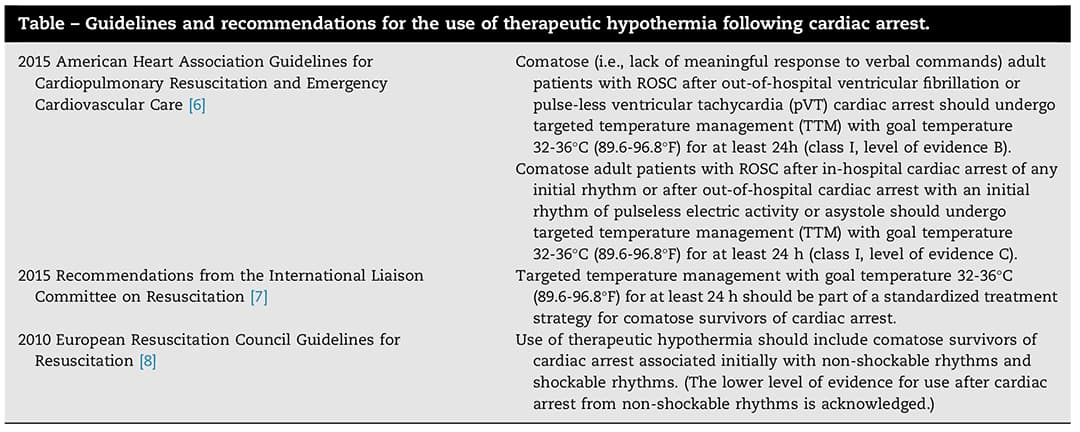
The following table lists some guidelines and recommendations for the use of TH following CA provided by the AHA, ILCOR, and the European Resuscitation Council Guidelines for Resuscitation:21
Initiation
In the initial randomized control trials demonstrating benefit with TH, target temperature was achieved within 4-8 h post-ROSC.12,3 However, the optimal timing for TH initiation as well as the time to goal target temperature remains uncertain.21 Mooney et al. demonstrated that among individuals undergoing TH post-CA (both shockable and non-shockable rhythms) there was a 20% increase in mortality for every hour that initiation of TH was delayed. Wolff et al. demonstrated that time to coldest temperature was an independent predictor of good neurologic outcome. A third study demonstrated that reaching target temperature within 6 h (early group) was associated with a greater likelihood of favorable neurologic outcome when compared with individuals reaching target temperature after 6 h (delayed group).10 In contrast, Haugk et al. found that shorter time to target temperature was associated with unfavorable neurologic outcomes (median duration 158 min-unfavorable versus 209 min-favorable). Based on the current data from observational studies as well as randomized control trials, the optimal timing to initiate TH and achieve target temperature remains unclear.21
Methods
There are multiple methods that have been documented for cooling a patient. Administration of cold intravenous fluids, ice packs around the patient, cold air blankets, and even spraying the patient with water and having a fan blowing on the patient. Another method is the use of intra-arrest transnasal evaporation.8 More controlled methods like a catheter being placed into a large central vein and temperature-controlled fluids being circulated through the catheter are in use. The cooled saline is never injected directly into the bloodstream but stays contained within the central catheter cooling the blood as it flows by the catheter.1
Although the intravascular catheter has demonstrated improved control of maintaining target temperature, given the lack of head-to-head comparison for clinical outcomes data, there does not appear to be one cooling method that is clearly superior21.
Temperature
Based on the original studies regarding TH, the AHA recommended 32-34°C as the target temperature for individuals undergoing TH post-ROSC following CA.21 In the setting of unknown optimal target temperatures for TH following CA, the Targeted Temperature Management (TTM) Trial Investigators recently published the largest randomized control trial of TH in patients who remained unconscious following ROSC after an OHCA.17 They randomized 950 patients post-CA to targeted temperature management at a target temperature of 33°C versus 36°C. At completion of the trial, there was no difference in mortality (50%-33°C, versus 48%-36°C, p=0.51) or poor neurologic outcome (52% for each group, p=0.87). Both groups were actively cooled as quickly as possible using ice-cold fluids, ice packs, and intravascular or surface temperature-management devices to maintain target temperature.17
It is possible that the degree of hypothermia may need to be titrated or adjusted to fit the severity of neurologic injury, and that there are likely sub-groups of patients who may derive benefit from a greater degree of hypothermia.21 The most recent ILCOR update on TH also favors continuing to target a temperature of 33°C, though acknowledging that 36°C can be used and will provide similar neurologic benefit.21
Sedation
TH could be an uncomfortable treatment modality. The patient should, during the treatment with hypothermia, be sedated until he or she is comfortable and without pain or risk of shivering.9 Adequate sedation and analgesia must be maintained throughout the hypothermia protocol until the patient has been rewarmed and has returned to normothermia and short-acting medications are preferred given the frequent reduced clearance of these medications in this patient population.21
Shivering is a physiologic reaction to cooling that frequently occurs when patients are between 35°C and 37°C and it should be treated because it slows the rate of cooling and increases metabolic activity.21
Rewarming
12–24 h after TH initiation rewarming should be initiated.21 Rewarming should occur slowly at a rate of 0.25–0.5°C per hour until the patient reaches normothermia.7 Rapid rewarming increases the risk of significant complications including hypotension from substantial vasodilation as well as hypoglycemia and hyperkalemia.21
Conclusion
Although TH has been part of clinical practice for over a decade and TTM now has class I recommendations from the AHA for use in all patients post ROSC, significant controversy and questions remain regarding several aspects of its implementation: (1) which patients post-CA should receive TH; (2) when should TH be initiated and how long should patients be cooled; (3) what is the ideal temperature for cooling, and should this decision be universally applied, or made on an individualized basis; and (4) how and when should we be making neurologic prognoses.21
References
- Alkadri ME, Peters MN, Katz MJ, White CJ. State-of-the-art paper: therapeutic hypothermia in out of hospital cardiac arrest survivors. Catheter Cardiovasc Interv. 2013;82:E482-90.
- Arrich J, Holzer M, Havel C, et al. Hypothermia for neuroprotection in adults after cardiopulmonary resuscitation. Cochrane Database Syst Rev. 2012;9:CD004128.
- Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, Gutteridge G, et al. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346:557-63.
- Bhattacharjee S, Baidya DK, Maitra S. Therapeutic hypothermia after cardiac arrest is not associated with favorable neurological outcome: a meta-analysis. Journal of Clinical Anesthesia. 2016;33:225-232.
- Booth CM, Boone RH, Tomlinson G, Detsky AS. Is this patient dead, vegetative, or severely neurologically impaired? Assessing outcome for comatose survivors of cardiac arrest. J Am Med Assoc. 2004;291:870-9.
- Brannstrom M, Niederbach C, Rodin AC. Experiences of surviving a cardiac arrest after therapeutic hypothermia treatment. An interview study. International Emergency Nursing. 2018;36:34-38.
- Callaway CW, Donnino MW, Fink EL, Geocadin RG, Golan E, Kern KB, et al.Part 8: Post-cardiac arrest care: 2015 American Heart Association guidelines update for cardio-pulmonary resuscitation and emergency cardiovascular care. Circulation. 2015;132(Suppl2): S465-82.
- Castrén M, Nordberg P, Svensson L, Taccone F, Vincent JL, Desruelles D, et al. Intra-arrest transnasal evaporative cooling: a randomized, prehospital, multicenter study (PRINCE: pre-ROSC intranasal cooling effectiveness). Circulation. 2010;122:729-36.
- Chiota NA, Freeman WD, Barrett K. Earlier hypothermia attainment is associated with improved outcomes after cardiac arrest. J Vasc Interv Neurol. 2011;4:14-7.
- Haugk M, Testori C, Sterz F, Uranitsch M, Holzer M, Behringer W, et al. Time to target temperature study group. Relationship between time to target temperature and outcome in patients treated with therapeutic hypothermia after cardiac arrest. Crit Care. 2011;15:R101.
- Hypothermia after Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. 2002;346:549-56
- Ketilsdottir A, Albertsdottir HR, Akadottir SH, Gunnarsdottir TH, Jonsdottir H. The experience of sudden cardiac arrest: becoming reawakened to life. Eur J Cardiovasc Nurs. 2013. https://dx.doi.org/10.1177/1474515113504864.
- Laver S, Farrow C, Turner D, Nolan J. Mode of death after admission to an intensive care unit following cardiac arrest. Intensive Care Med. 2004;30:2126-8.
- McNally B, Robb R, Mehta M, et al, Centers for Disease Control and Prevention. Out-of-hospital cardiac arrest surveillance—Cardiac Arrest Registry to Enhance Survival (CARES), United States, October 1, 2005-December 31, 2010. MMWR Surveill Summ. 2011;60:1-19.
- Mooney MR, Unger BT, Boland LL, Burke MN, Kebed KY, Graham KJ, et al. Therapeutic hypothermia after out-of-hospital cardiac arrest: evaluation of a regional system to increase access to cooling. Circulation. 2011;124:206-14.
- Nielsen N, Wetterslev J, Cronberg T, Erlinge D, Gasche Y, Hassager C, et al. Targeted temperature management at 33°C versus 36°C after cardiac arrest. N Engl J Med. 2013;369:2197-206.
- Park JH, Wee JH, Choi SP, Oh JH, Cheol S. Assessment of serum biomarkers and coagulation/fibrinolysis markers for prediction of neurological outcomes of out of cardiac arrest patients treated with therapeutic hypothermia. Clinical and Experimental Emergency Medicine. 2019 https://doi.org/10.15441/ceem.17.273.
- Peberdy MA, Callaway CW, Neumar RW, et al. Post-cardiac arrest care: 2010 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2010;122(Suppl3):S768-86.
- Samaniego EA, Persoon S, Wijman CA. Prognosis after cardiac arrest and hypothermia: a new paradigm. Curr Neurol Neurosci Rep. 2011;11:111-9.
- Silverman MG, Scirica BM. Cardiac arrest and therapeutic hypothermia. Trends in Cardiovascular Medicine. 2016;26:337-344.
- Swedish Council of CPR. http://www.hlr.nu/fakta-riktlinjer/. 2011. Accessed August 12, 2019.
- Van Lommel P. Near-death experiences: the experience of the self as real and not as an illusion. Ann N Y Acad Sci. 2011;1234:19-28.
- Wolff B, Machill K, Schumacher D, SchulzkiI, Werner D. Early achievement of mild therapeutic hypothermia and the neurologic outcome after cardiac arrest. IntJCardiol. 2009;133:223-8.