Influenza: Types, Treatment, and Prevention
 Influenza Overview
Influenza Overview
The influenza virus is responsible for the morbidity and mortality of millions of people every year throughout the world. The World Health Organization (WHO) has recently warned that influenza is a global health threat and has the potential to become a global pandemic unless nations take measures to “prevent, control and prepare.”13
There have been four major influenza outbreaks since the H1N1 flu pandemic in 1918 that killed 50 million people, including 675,000 in the United States.5 In 1957, 1 million people died from the H2N2 virus, and another 1 million died in 1968 from the H3N2 virus.5 The last pandemic occurred in 2009 when the H1N1 virus killed less than 0.3 million people in the first year.5
Since 1918, the death toll has decreased with each pandemic. This has resulted from widespread use of the flu vaccine, better treatment options once infected, and an overall improved knowledge base of the disease. The Center for Disease Control’s (CDC) National Stockpile manages and distributes medical countermeasures, including antiviral medications, ventilators, and personal protective equipment, as well as other life-saving pharmaceuticals and medical supplies.11 While advances in medical care, continuing medical education, and countermeasures contributed to a reduction in deaths in the past three pandemics, and although these measures are widely available today, a novel influenza A virus could abruptly change to become a more human-adapted virus, spreading efficiently from person to person and causing significant morbidity and mortality worldwide.11
Types of Influenza
There are four types of influenza viruses: A, B, C, and D, of which the human influenza A and B viruses cause seasonal epidemics of disease (known as the flu season) almost every winter in the United States.7 Influenza A viruses are the only influenza viruses known to cause flu pandemics.7 Influenza type C infections generally cause mild illness and are not thought to cause human flu epidemics while Influenza D viruses primarily affect cattle and are not known to infect or cause illness in people.
Every year many individuals in multiple communities complain that the current year’s flu vaccine is not effective. Taking a deeper dive into the actual science behind influenza shows how complicated it can be to determine which virus may be the one to cause the most significant damage from year to year. The following is from the CDC website and explains the subtypes of influenza viruses, which are based on surface proteins located on the virus:
Influenza A viruses are divided into subtypes based on two proteins on the surface of the virus: hemagglutinin (H) and neuraminidase (N). There are 18 different hemagglutinin subtypes and 11 different neuraminidase subtypes (H1 through H18 and N1 through N11, respectively). While there are potentially 198 different influenza A subtype combinations, only 131 subtypes have been detected in nature. Current subtypes of influenza A viruses that routinely circulate in people include: A(H1N1) and A(H3N2). Influenza A subtypes can be further broken down into different genetic “clades” and “sub-clades.” See the “Influenza Viruses” graphic below for a visual depiction of these classifications.7
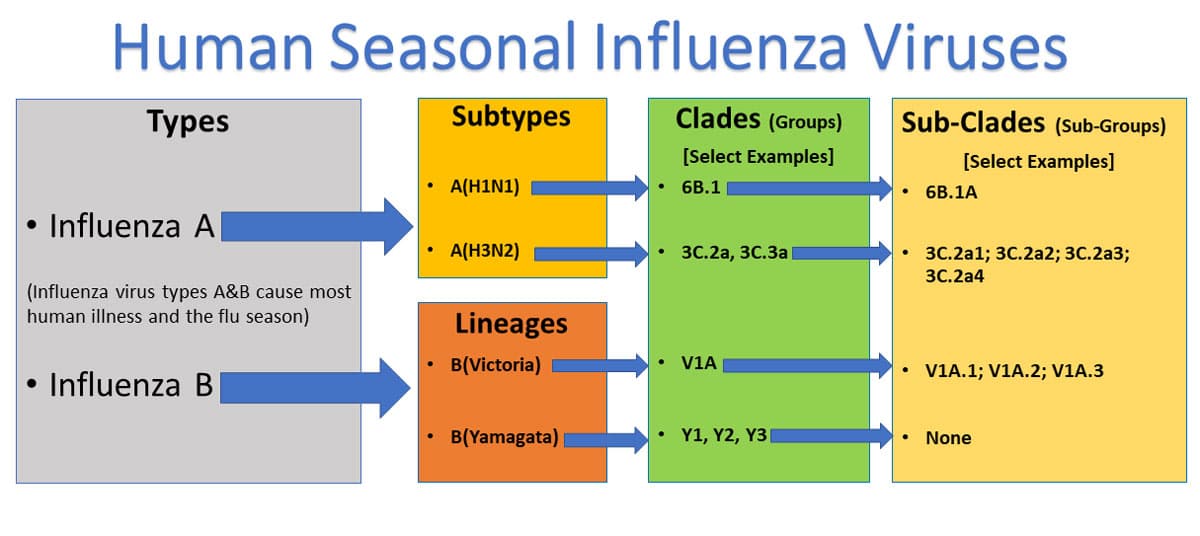
From the information above, one can see where the more common H1N1 virus falls in regards to the Influenza Types A and B.
Diagnosis
Influenza diagnosis is dependent on either identification of virus or the detection of viral protein/viral RNA in respiratory tract secretions.12 Rapid influenza diagnostic tests (RIDTs) utilizing viral antigen detection are commonly performed in the outpatient setting because results are obtained quickly, usually before the end of the visit.2 According to Jester, these can be completed in 10-20 minutes.11 However, these tests have a lower sensitivity (50%-70%) than other currently available tests.12 Reverse transcriptase polymerase chain reaction (RT-PCR) and rapid molecular assays are two other testing modalities with improved sensitivity (86%-100% and 66%-100%, respectively) and are available as Clinical Laboratory Improvement Amendments (CLIA)—waived tests that can be used at point of care. The rapid molecular assays are more accurate than immunoassays and can yield results in 15 to 30 minutes.3
The CDC and a subset of public health laboratories routinely perform next-generation genetic sequencing on all respiratory specimens received.11 The CDC and state health departments use these virologic data to monitor influenza virus characteristics, disease activity, geographic spread, virulence, antiviral effectiveness, and vaccine effectiveness, as well as to develop candidate vaccine viruses.4
Prevention
Influenza vaccination is now considered the first and most important step in protecting against influenza viruses.11 One influenza A (H1N1), one influenza A (H3N2), and one or two influenza B viruses (depending on the vaccine) are included in each season’s influenza vaccines.5
The first clinical trials of influenza vaccines began in the 1930s, and, by 1942, large studies of inactivated influenza vaccines were underway.1 For the 2017–2018 influenza season, multiple influenza vaccine formulations were available, including standard dose, high dose, adjuvanted, and two more recently developed vaccines made without eggs: a cell-based manufactured vaccine and a recombinant protein vaccine.11 These latter two vaccines represent new technologies that may allow for faster vaccine production in the event of a pandemic.11 Over the 14 flu seasons from 2004 to 2018, the estimated influenza vaccination effectiveness ranged from 10% to 60% with effectiveness greater than 40% 9 times.6
Treatment
The CDC provides frequent updates for prophylaxis and treatment based on circulating strains and resistance patterns, available at https://www.cdc.gov/flu/professionals/antivirals/index.htm.
A class of antiviral drugs called neuraminidase inhibitors were first approved in 1999. The goals of an antiviral medication are to lessen the duration of influenza symptoms and attempt to prevent serious complications, especially when treatment is started early after illness onset. Oseltamivir (Tamiflu), zanamivir (Relenza), and peramivir (Rapivab) are neuraminidase inhibitors that are currently available in the United States.2 Oseltamivir and zanamivir are available orally and are active against both influenza A and B viruses while Peramivir is an intravenous (IV) formulation and indicated in patients aged older than 2 years with symptoms for fewer than 2 days. The CDC recommended three US Food and Drug Administration–approved antiviral medications for treatment of influenza during the 2017–2018 season.8
In a study published in the British Journal of Medicine, treatment with a neuramidase inhibitor decreased symptom duration by approximately one day, decreased associated otitis media, and decreased prescription of unnecessary antibiotics.10 In one study of children aged 1 to 3 years, treatment within 24 hours with oseltamivir decreased symptom duration by three days.9 The arsenal of antimicrobial drugs is effective only when a secondary bacterial infection needs to be treated.
Prevention, diagnosis, and treatment of influenza has drastically improved since the 1918 pandemic. That being said, health care providers need to stay educated and alert to local and regional outbreaks to help prevent themselves from getting the disease but to also know how to detect and treat the infection in those they care for.
For more information about how to protect yourself as a provider against infectious diseases, check out our continuing education courses. Browse CareerCert’s accredited online courses today!
References
- Barberis I, Myles P, Ault SK, Bragazzi NL, Martini M. History and evolution of influenza control through vaccination: from the first monovalent vaccine to universal vaccines. J Prev Med Hyg. 2016; 57(3):E115–E120.
- Bass PF. Influenza and RSV: How to suspect, diagnose, treat. 2017. http://contemporarypediatrics.com. Accessed January 11, 2020.
- Belshe RB, Coelingh K, Ambrose CS, Woo JC, Wu X. Efficacy of live attenuated influenza vaccine in children against influenza B viruses by lineage and antigenic similarity. Vaccine. 2010;28(9):2149-2156.
- Centers for Disease Control and Prevention. Overview of influenza surveillance in the United States. 2018. https://www.cdc.gov/flu/weekly/overview.htm. Accessed January 15, 2020.
- Centers for Disease Control and Prevention. Pandemic influenza. 2017. https://www.cdc.gov/flu/pandemic-resources. Accessed January 15, 2020.
- Centers for Disease Control and Prevention. Seasonal influenza vaccine effectiveness, 2005-2018. https://www.cdc.gov/flu/vaccines-work/effectiveness-studies.htm. Updated February 15, 2018. Accessed January 10, 2020.
- Center for Disease Control and Prevention. Types of viruses. 2019. https://www.cdc.gov/flu/about/viruses/types.htm. Accessed January 12, 2020.
- Centers for Disease Control and Prevention. What you should know about flu antiviral drugs. 2018. https://www.cdc.gov/flu/antivirals/whatyoushould.htm. Accessed January 19, 2020.
- Heinonen S, Silvennoinen H, Lehtinen P, et al. Early oseltamivir treatment of influenza in children 1-3 years of age: a randomized controlled trial. Clin Infect Dis. 2010;51(8):887-894.
- Jefferson T, Jones M, Doshi P, Spencer EA, Onakpoya I, Heneghan CJ. Oseltamivir for influenza in adults and children: systematic review of clinical study reports and summary of regulatory comments. BMJ. 2014;348:g2545.
- Jester BJ, Uyeki TM, Patel A, Koonin L, Jernigan DB. 100 Years of Medical Countermeasures and Pandemic Influenza Preparedness. American Journal of Public Health. 2018;108:1469-1472.
- Merckx J, Wali R, Schiller I, et al. Diagnostic accuracy of novel and traditional rapid tests for influenza infection compared with reverse transcriptase polymerase chain reaction: a systematic review and meta-analysis. Ann Intern Med. 2017;167(6):394-409.
- World Health Organization. Global influenza strategy 2019–2030. 2019. https://tinyurl.com/yywr384q. Accessed January 10, 2020).
Dan Bunker DNAP, MSNA, CRNA—Dan has worked in the healthcare industry for nearly 30 years. He worked as a registered nurse in the coronary care ICU for 7 years and was a flight nurse with Intermountain’s Life Flight for nearly 10 years. He has been a certified registered nurse anesthetist (CRNA) for 11 years, working in the hospital setting as well as maintaining his own private practice. In addition, he is a professor in the nurse anesthesia program at Westminster College in Salt Lake City, Utah. He has served in various leadership roles within the Utah Association of Nurse Anesthetists (UANA) and is currently the president-elect.