Coronavirus: Vaccine, Prevention, and the Latest Information
Overview
Coronaviruses (CoV) are positive RNA-enveloped viruses (Ori). The first two human coronaviruses, HCoV-229E and HCoV-OC43, were discovered in the mid-1960s (Ori). Coronaviruses are a well-known cause of the common cold (Geller). According to the World Health Organization (WHO), coronaviruses are a large family of viruses that cause illness ranging from the common cold to more severe diseases such as severe acute respiratory syndrome (SARS-CoV) and Middle East respiratory syndrome (MERS-CoV). Two recent large outbreaks of HCoV were in 2002 and 2012. The 2002 HCoV epidemic was severe acute respiratory syndrome human coronavirus (SARS-HCoV), and in 2012 the epidemic was located in the Middle East and was identified as MERS-HCov (Zaki). Coronaviruses are a large family of viruses that are common in humans and many different species of animals, including camels, cattle, cats, and bats (CDC).
The definition of a novel coronavirus (nCoV) is one that has not been found in humans before that point. In November of 2019, a nCoV was found in Wuhan, China. That coronavirus was first labeled as 2019-nCoV.
Following WHO best practices for naming of new human infectious diseases, which were developed in consultation and collaboration with the World Organization for Animal Health (OIE) and the Food and Agriculture Organization of the United Nations (FAO), WHO has named the disease COVID-19, short for “coronavirus disease 2019” (WHO-situation report). Both names will be used throughout this article.
COVID-19 is not the same as other coronaviruses that commonly circulate among humans and cause mild illness, like the common cold. Some cause illness in people, and others, such as canine and feline coronaviruses, only infect animals. Animal coronaviruses rarely infect people and spread between people, which is suspected to have occurred for the virus that causes COVID-19. MERS and SARS are two other examples of coronaviruses that originated from animals and then spread to people.
Diagnosis
Currently, confirmation of 2019-nCoV infection is performed at CDC using the CDC real-time RT-PCR assay for 2019-nCoV on respiratory specimens (which can include nasopharyngeal or oropharyngeal aspirates or washes, nasopharyngeal or oropharyngeal swabs, bronchoalveolar lavage, tracheal aspirates, or sputum) and serum (CDC). The CDC is the only location in the United States that can confirm the diagnosis of nCoV at this time.
Symptoms
The Centers for Disease Control and Prevention (CDC) stated that patients have reportedly had mild to severe respiratory illness.
More common symptoms for 2019-nCoV:
- Fever (83-98%)
- Cough (76-82%)
- Myalgia or Fatigue (11-44%)
- Shortness of Breath
- Sore Throat
Less common symptoms of 2019-nCoV:
- Sputum production
- Headache
- Hemoptysis
- Diarrhea
The incubation period is estimated at ~5 days (95% confidence interval, 4 to 7 days) (CDC).
The severe problem associated with the virus is pneumonia in bilateral lungs which can lead to respiratory failure and death. The following is clinical course information provided by the CDC:
Acute respiratory distress syndrome (ARDS) developed in 17–29% of hospitalized patients, and secondary infection developed in 10%. Between 23–32% of hospitalized patients with 2019-nCoV infection required intensive care for respiratory support. Some hospitalized patients have required advanced organ support with invasive mechanical ventilation (4–10%), and a small proportion have also required extracorporeal membrane oxygenation (ECMO, 3–5%). Other reported complications include acute cardiac injury (12%) and acute kidney injury (4–7%). Among hospitalized patients with pneumonia, the case fatality proportion has been reported as high as 11–15%. However, as this estimate includes only-hospitalized patients, and therefore is biased upward.
No vaccine or specific treatment for 2019-nCoV infection is available; care is supportive (CDC).
Health Care Professionals
The CDC has listed recommendations for health care professionals to follow if they believe an individual is a Persons Under Investigation (PUI) for 2019-nCoV. The criteria are intended to serve as guidance for evaluation. Patients should be evaluated and discussed with public health departments on a case-by-case basis if their clinical presentation or exposure history is equivocal (e.g. uncertain travel or exposure).
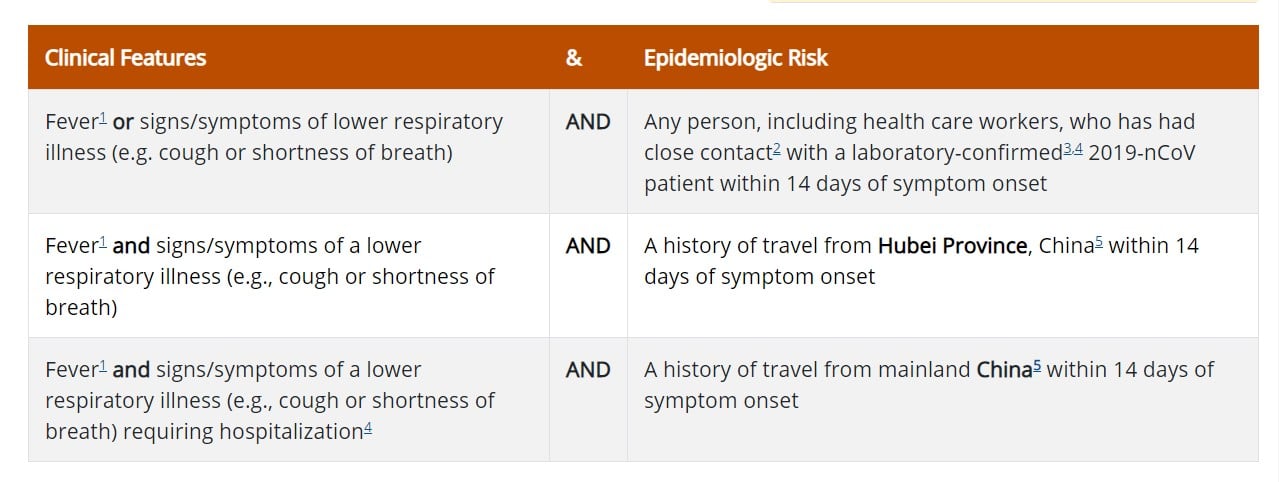
-
Fever may be subjective or confirmed
-
Close contact is defined as—
-
being within approximately 6 feet (2 meters) of a 2019-nCoV case for a prolonged period of time; close contact can occur while caring for, living with, visiting, or sharing a health care waiting area or room with a 2019-nCoV case – or –
-
having direct contact with infectious secretions of a 2019-nCoV case (e.g., being coughed on). If such contact occurs while not wearing recommended personal protective equipment or PPE (e.g., gowns, gloves, NIOSH-certified disposable N95 respirator, eye protection), criteria for PUI consideration are met
-
-
Documentation of laboratory-confirmation of 2019-nCoV may not be possible for travelers or persons caring for patients in other countries.
-
Category also includes any member of a cluster of patients with severe acute lower respiratory illness (e.g., pneumonia, ARDS) of unknown etiology in which 2019-nCoV is being considered that requires hospitalization. Such persons should be evaluated in consultation with state and local health departments regardless of travel history.
-
For persons with travel to China within 14 days that are being regularly monitored by local health departments or referred for evaluation from border screening, testing for nCoV can be considered at the discretion of the health officials for all persons with illnesses with fever and lower respiratory symptoms (those hospitalized and those not hospitalized).
Healthcare providers should immediately notify both infection control personnel at their healthcare facility and their local or state health department in the event of a PUI for 2019-nCoV (CDC).
Prevention
As listed earlier, there is no vaccine currently available for 2019-nCov, and treatment is supportive.
The following is a list of guidelines from the CDC to help in the prevention of spreading COVID-19, and other diseases that can be spread by person to person contact:
- Avoid close contact with people who are sick.
- Avoid touching your eyes, nose, and mouth.
- Stay home when you are sick.
- Cover your cough or sneeze with a tissue, then throw the tissue in the trash.
- Clean and disinfect frequently touched objects and surfaces using a regular household cleaning spray or wipe.
- Follow CDC’s recommendations for using a facemask.
- CDC does not recommend that people who are well wear a facemask to protect themselves from respiratory diseases, including COVID-19.
- Facemasks should be used by people who show symptoms of COVID-19 to help prevent the spread of the disease to others. The use of facemasks is also crucial for health workers and people who are taking care of someone in close settings (at home or in a healthcare facility).
- Wash your hands often with soap and water for at least 20 seconds, especially after going to the bathroom; before eating; and after blowing your nose, coughing, or sneezing.
- If soap and water are not readily available, use an alcohol-based hand sanitizer with at least 60% alcohol. Always wash hands with soap and water if hands are visibly dirty.
Vaccine
Alex Azar, U.S. Secretary of Health and Human Services and the White House COVID-19 response coordinator, stated on March 9, 2020, that “rapid work is being done on a vaccine that should go into clinical trials very soon.” He also reported that “we have a therapeutic already in clinical trials.” That being said, an article published in The Journal of the American Medical Association in 2018 stated that—in the development of vaccines for emerging infectious diseases with pandemic potential—the traditional approach of isolating and growing a pathogen does not support an effective response to these continually emerging threats (Graham). The article emphasizes that it will be critical to exploit modern-day technological advances, pre- emptively establish detailed information on each family of viral pathogens, and invest in more infrastructure for surveillance in developing countries to expedite pathogen identification and jumpstart the process of vaccine development (Graham).
Treatment
Clinical management includes prompt implementation of recommended infection prevention and control measures and supportive management of complications, including advanced organ support if indicated (CDC). Healthcare personnel should care for patients in an airborne infection isolation room (AIIR) and standard precautions, contact precautions, and airborne precautions and eye protection should be used when caring for the patient (CDC).
The decision to monitor a patient in the inpatient or outpatient setting should be made on a case-by-case basis and this decision will depend not only on the clinical presentation but also on the patient’s ability to engage in monitoring and the risk of transmission in the patient’s home environment (CDC).
The WHO has developed a special tool for determining which diseases and pathogens to prioritize for research and development in public health emergency contexts (WHO). This tool is used to help pinpoint which diseases may cause a public health risk because of their potential to cause an epidemic and for which there are no countermeasures, or the measures in place are insufficient (WHO). The latest list was released in 2018 and will be updated soon. The list is not exhaustive, nor does it have any information on which disease may be the most likely to cause an epidemic. The following is a list of diseases released by the WHO:
- Crimean-Congo haemorrhagic fever (CCHF)
- Ebola virus disease and Marburg virus disease
- Lassa fever
- Middle East respiratory syndrome coronavirus (MERS-CoV) and severe acute respiratory syndrome (SARS)
- Nipah and henipaviral diseases
- Rift Valley fever (RVF)
- Zika
- Disease X (Disease X represents the knowledge that a serious international epidemic could be caused by a pathogen currently unknown to cause human disease, and so the R&D Blueprint explicitly seeks to enable cross-cutting R&D preparedness that is also relevant for an unknown “Disease X” as far as possible.)
Coronavirus Compared to Influenza
Coronavirus is making headlines around the globe. To date, there have been more than 1,210, 000 confirmed illnesses and 67,500 deaths due to the coronavirus (WHO). This year in the United States alone influenza caused an estimated 19 million illnesses, 180,000 hospitalizations, and 10,000 deaths (CDC).
The measure scientists use to determine how easily a virus spreads is known as the “basic reproduction number,” or R0 (pronounced R-nought). R0 is an estimate of the average number of people who catch the virus from a single infected person. Comparing R0 between influenza and 2019-nCoV helps determine the risk of infection. The R0 for the influenza virus is 1.3 (Sheikh). The R0 for 2019-nCoV is still being determined by researchers. According to a recent study in The New England Journal of Medicine, in its early stages the epidemic doubled in size every 7.4 days (Li). With a mean serial interval of 7.5 days, the R0 was estimated to be 2.2 (Li). This indicates that 2019-nCoV is roughly twice as contagious as influenza.
Latest Information
For up to date information on COVID-19, readers are encouraged to visit the Centers for Disease Control and Prevention at www.cdc.gov and the World Health Organization at www.who.int.
References
Center for Disease Control and Prevention. Coronavirus. https://www.cdc.gov. Accessed March 9, 2020.
Coronavirus: Everything you need to know. Fox News. https://www.foxnews.com/health/coronavirus-everything-you-need-to-know. Accessed March 9, 2020.
Geller C, Varbanov M, Duval RE. Human coronaviruses: insights into environmental resistance and its influence on the development of new antiseptic strategies. Viruses. 2012;4:3044–3068.
Graham BS, Mascola JR, Fauci AS. Novel Vaccine Technologies. Essential components of an adequate response to emerging viral diseases. JAMA. Published online March 22, 2018. doi:10.1001/jama.2018.0345.
Li Q, Med M, Guan X, Wu P, Wang X, Zhou L, Tong Y, Ren R, Leung K, Lau EHY, Wong JY, Xing X, Xiang N, et al. Early Transmission Dynamics in Wuhan, China, of Coronavirus-Infected Pneumonia. The New England Journal of Medicine. 2020; DOI: 10.1056/NEJMoa2001316.
Ori G, Shemer Y, Fuchs L, Ferster OA, Almog Y. . American Journal of Respiratory and Critical Care Medicine. 2016; 193(1).
Rettner R. How does the new coronavirus compare with the flu?. February 7, 2020. Scientific American. https://www.scientificamerican.com/article/how-does-the-new-coronavirus-compare-with-the-flu/. Accessed February 11, 2020.
Sheikh k, Watkins D, Wu J, Grondahl M. How bad will the coronavirus outbreak get? Here are 6 key factors. The New York Times. February 7, 2020. https://www.nytimes.com/interactive/2020/world/asia/china-coronavirus-contain.html. Accessed February 11, 2020.
World Health Organization. Coronaviruses. https://www.who.int/health-topics/coronavirus. Accessed February 6, 2020.
World Health Organization. Prioritizing diseases for research and development in emergency contexts. https://www.who.int/activities/prioritizing-diseases-for-research-and-development-in-emergency-contexts. Accessed March 9, 2020.
World Health Organization. Situation Report- 56. https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200316-sitrep-56-covid-19.pdf?sfvrsn=9fda7db2_2. Accessed March 17, 2020.
Zaki AM, van Boheemen S, Bestebroer TM, Osterhaus AD, Fouchier RA. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med. 2012;367:1814–1820.
Dan Bunker DNAP, MSNA, CRNA—Dan has worked in the healthcare industry for nearly 30 years. He worked as a registered nurse in the coronary care ICU for 7 years and was a flight nurse with Intermountain’s Life Flight for nearly 10 years. He has been a certified registered nurse anesthetist (CRNA) for 11 years, working in the hospital setting as well as maintaining his own private practice. In addition, he is a professor in the nurse anesthesia program at Westminster College in Salt Lake City, Utah. He has served in various leadership roles within the Utah Association of Nurse Anesthetists (UANA) and is currently the president-elect.




